If you search online for how the nucleus was discovered you will probably find a description of Rutherford’s gold foil experiment, where a beam of alpha particles hit a piece of gold and then the position of the alpha particle is found on a screen that is wrapped around the gold. Now, an experiment like this was done in 1913.
However, it wasn’t done by Rutherford nor did it inspire Rutherford to discover the nucleus! In fact, the 360-degree experiment was actually conducted to verify Rutherford’s ideas about the nucleus that he published two years earlier. The real influential experiment was done in 1909 and it involved eight different metals(not just gold) but was far simpler. So simple, in fact, that Rutherford gave the experiment to an undergrad as busywork.
Table of Contents
Rutherford and William Bragg’s Theory
How Scintillation was Discovered by Hans Geiger and Ernest Rutherford
Continuation of the Experiment with Marsden
Why the Nuclear Model Wasn’t Inspired by the Gold Foil Experiment
References
Rutherford and William Bragg’s Theory

It all started in 1899. That was when Rutherford, who was a graduate student at Cambridge, noticed that the radiation from uranium was “complex” in that some was easy to block and some wasn’t. He labeled the easiest blocked radiation as alpha and the other radiation as beta.
Rutherford then got a job at McGill University in Montreal, where he had, as he put it, “a swell lab” and he soon attracted a large group of collaborators and students and proceeded to do a lot of amazing work that I talked about in my last video.
In 1904, an Australian named William Bragg published a theory that alpha particles travel through thin solids without any deviation. In general, Rutherford thought very highly of Bragg’s work but he disagreed on this one point. See, Rutherford believed that all matter was filled with electrons or teeny tiny charged particles, which created relatively strong electrical forces as they are so small you can get really close to them.
J.J. Thomson, Rutherford’s advisor at Cambridge, had discovered electrons (which he called corpuscles) back in 1897 and by 1904 Thomson concluded that, “atoms consist of a number of negatively electrified corpuscles enclosed in a sphere of uniform positive electrification.[1]”
This was colloquially called the “plum pudding” model of atoms, where the electrons were the raisons (or if you are a Brit, the plums) in a positive pudding. Around this time Rutherford became convinced that alpha particles were positive, had twice the charge of an electron and around 7,000 times the mass.
Despite this, Rutherford thought that the light electrons in thin metal could push heavy alpha particles, just not by much. In 1906, Rutherford published an experimental proof of his theory. He put a small piece of radioactive wire in a groove and then had the beam of alpha particles from the wire go through a small slit and illuminate a photographic plate.
He then covered half of the slit with a mica screen. The beam that went through the mica was slightly more spread out then the beam that did not go through the mica. In fact, the thin mica caused the beam to spread out by about 2 percent[2]. Rutherford concluded, “Such a result brings out clearly the fact that the atoms of matter must be the seat of very intense electrical forces – a deduction in harmony with the electronic theory of matter.[3]”
How Scintillation was Discovered by Hans Geiger and Ernest Rutherford

In 1907, Rutherford, who felt left out in the “colonies” of Canada, got a job at the University of Manchester where he met a young German scientist named Hans Geiger. Geiger and Rutherford then determined that a screen made of phosphorescent zinc sulphide would glow when hit with alpha particles, and only alpha particles, and could be studied with a microscope (called scintillation).
Rutherford hated scintillation and left it to the younger, and more patient, Geiger, writing a friend that, “Geiger is a demon at the work of counting scintillations and could count at intervals for a whole night without destroying his equanimity. I damned vigorously and retired after two minutes[4].”
There was another problem with scintillation, “The doubt,” as Rutherford put it, “however, at once arises whether every alpha particle produces a scintillation[5].” Therefore, Rutherford turned to “an electric method”. Now an alpha particle carries a charge of 2e or twice the charge on a single electron, which is very, very small, way too small for an electrometer.
Luckily, Rutherford’s friend from his time with JJ Thomson, John Townsend, had found that if he put a gas at low pressure under really high voltage until it almost sparked, just a little charge would create an avalanche of charges so charges could be magnified. By January of 1908, Rutherford wrote that, “Under such conditions, the ionization produced in the gas by the alpha particle is magnified 2000 times by collision.
The effect of each alpha particle is marked enough to show an audience.[6]” By July, Rutherford and Geiger published their conclusion that, “the number of scintillations…. is, within the limit of experimental error, equal to the number of alpha particles falling upon it, as counted by the electric method.[7]”
At around the same time as the electric experiment, Geiger was also studying the number of alpha particles that were emitted by a substance by scintillations when he noticed it was scattered by air. Geiger mentioned it to Rutherford and Rutherford recalled his experiment with the mica and suggested that Geiger should examine exactly how alpha particles diverted when going through thin solids with the scintillation method.
Geiger thus put radium in a small lead evacuated cone with one end covered in thin mica that let the alpha particles escape. He then had the beam of particles go through a small slit that could be covered with foil and then examined the alpha particles that hit a phosphorescent screen with a microscope that could be moved up or down by about 10 mm.
In this way Geiger produced smooth curves of the position of alpha particles diverted by thin metals.
Marsden and Rutherford
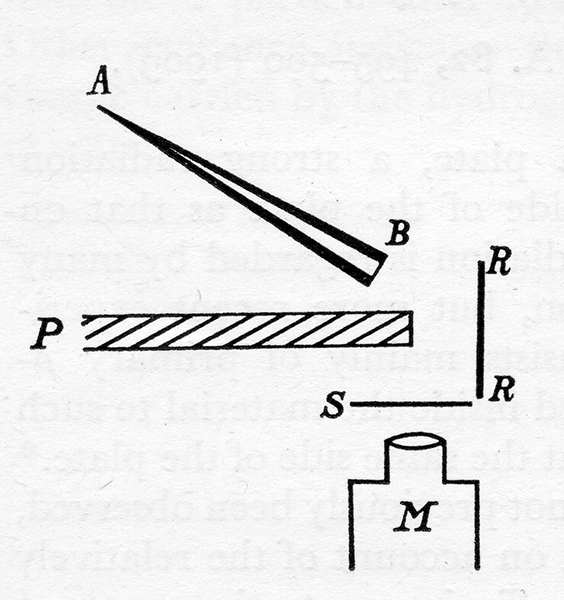
The next year, a 20-year-old undergrad named Ernest Marsden joined the research group. What happened next is best described by Rutherford himself, “One day Geiger came to me and said, ‘Don’t you think that young Marsden…ought to begin a small research?’ Now I had thought that too, so I said, ‘Why not let him see if any a-particles can be scattered through a large angle?’ I may tell you in confidence that I did not believe that they would be, since we knew that the a-particle was a very fast massive particle, with a great deal of energy…
Then I remember two or three days later Geiger coming to me in great excitement and saying, ‘We have been able to get some of the a-particles coming backwards.’[8]” So, what did Marsden do? Well, he used the same cone of radioactive material as before but had it hit a foil of metal at an angle and then have the reflected alpha particles hit a screen that was examined by a microscope.
He also placed a lead barrier between the screen and the radioactive source so that everything he saw had to reflect off the foil. Geiger and Marsden then experimented with eight different metals and found that a few alpha particles would bounce off thin metals, where the heavier the metal, the more alpha particles would reflect. By the way, this is not to imply that a lot of alpha particles reflect. In fact, in they found that only about one in 8,000 alpha particles would reflect of a piece of platinum![9]
Still, they couldn’t figure out why any heavy alpha particles would bounce off a thin metal foil, it didn’t make sense. Rutherford recalled, “It was quite the most incredible event that has ever happened to me in my life. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you.[10]”
Rutherford thought about it for over a year, and by December of 1910 wrote a friend, “I think I can devise an atom much superior to J.J.’s… It will account for the reflected alpha particles observed by Geiger and generally I think will make a fine working hypothesis.[11]”
Rutherford recalled that, “I realized that this scattering backwards must be the result of a single collision, and when I made calculations I saw that it was impossible to get anything of that order of magnitude unless you took a system in which the greater part of the mass of the atom was concentrated in a minute nucleus.[12]”
Nuclear Model Wasn’t Inspired by the Gold Foil Experiment

On March 7, 1911, Rutherford published an article where he assumed that an atom contained, “a positive charge Ne at its center, and surrounded by a distribution of negative electricity Ne uniformly distributed within a sphere of radius R.[13]” Rutherford then derived from the experimental results that the charge on the platinum nucleus was about 100e and the nucleus has a radius that is more than 10,000 times smaller than the atom[14].
He was startlingly close, we now think that platinum has a charge 78e and the nucleus has a radius around 100,000 times smaller than the atom!
The nucleus is ridiculously small, “if an atom were expanded to the size of a cathedral, the nucleus would be only about the size of a fly.[15]” A fly that holds 99.9997% of the weight! Or, in reverse, 99.999999999999999% of an atom is filled with only 0.0003% of the weight.
So, if everything is basically almost nothing then why are solid objects solid? Why don’t I just fall through the floor? Well, it is because of the teeny tiny electrons in everything. The electrons in the atoms in your shoes push against the electrons in the atoms in the floor when they get close enough to each other.
You are not really touching the floor! You are actually hovering above it due to electrical forces! Crazy, eh?
In 1913, Geiger and Marsden did conduct a gold foil experiment although it wasn’t done with a wrap around screen but a microscope coated with a screen that could be moved in a full circle. That experiment validated Rutherford’s conclusions and proved that the nucleus was positive, which he was pretty confident about in 1910 but it was nice to have validation.
Rutherford’s model created a lot of interesting questions. One of the biggest ones had to do with the charges of the negative electrons and the positive nucleus. Opposite charges attract, so what keeps the negative electrons from being sucked into the positive nucleus?
Even if the electrons were spinning around the nucleus like planets then they would be accelerating charges (spinning is a form of acceleration), which should, according to Maxwell’s laws, be creating electromagnetic waves, which would cause the electrons to lose energy and spiral into the nucleus.
In other words, according to the laws of classical physics, Rutherford’s atom should just implode. But we exist. So, either Rutherford was wrong or the basic rules of physics don’t work for atoms! This is why, in July of 1913, a young Danish man who was working for Rutherford named Neil Bohr made the first quantum mechanical view of the atom, the Bohr model.
In this model, Bohr relied on the results from a 1900 paper on blackbody radiation written by a man named Max Planck. But to tell that story (and the story of quantum mechanics in general), I first want to take a step back to the man who came up with the idea of blackbody radiation in the first place. This is a fabulous story of a tiny man and his large friend who transformed our world with a prism and literally got gold from the sun. How Kirchhoff and Bunsen discovered spectroscopy, the systematic study of the light from elements, and how that led to the idea of blackbody radiation is next time on the lightning tamers.
References
[1] Thomson, JJ “On the Structure of the Atom…” The London, Edinburgh and Dublin Phil. Mag vol. 7 1904
[2] Rutherford, E “Retardation of the alpha Particle from Radium in passing through Matter” The London, Edinburgh and Dublin Phil. Mag vol. 12 1906 p. 144
[3] Rutherford, E “Retardation of the alpha Particle from Radium in passing through Matter” The London, Edinburgh and Dublin Phil. Mag vol. 12 1906 p. 145
[4] Rutherford to Bumstead July 11, 1908 quoted in Eve, A. S. Rutherford p. 180
[5] Rutherford, E and Geiger, H “An Electrical Method of Counting the Number of alpha particles from Radio-active Substances” Proceedings of the Royal Society of London. Vol. 81 July 17, 1908 p. 141
[6] Rutherford, E to Hahn, O, Jan 27, 1908 quoted in Eve, A.S. Rutherford p. 175
[7] Rutherford, E and Geiger, H “An Electrical Method of Counting the Number of alpha particles from Radio-active Substances” Proceedings of the Royal Society of London. Vol. 81 July 17, 1908 p. 161
[8] Rutherford, Ernest “Forty Years of Physics” in Background to Modern Science, 1938 p 48
[9] Geiger, H, Marsden, E “On a Diffuse Reflection of the alpha Particles” Royal Society of London vol. ? June 17, 1909 p. 499
[10] Rutherford, Ernest “Forty Years of Physics” in Background to Modern Science, 1938 p 48
[11] Rutherford, E. to Boltwood, B, December 1910, recalled in Geiger, Reeves, Richard A Force of Nature: The Frontier Genius of Ernest Rutherford p. 80
[12] Rutherford, Ernest “Forty Years of Physics” in Background to Modern Science, 1938 p 48
[13] Rutherford, E “The Scattering of alpha and beta particles by Matter and the Structure of the Atom” Philosophical Magazine vol. 21 (1911) p. 671
[14] The actual quote was “We shall suppose that for distances less than 10-12 cm the central charge… may be supposed to be concentrated at a point” However, he also assumed that the atom had a radius of about 10-8 cm which makes the nucleus about 10,000 times smaller than the atom. Rutherford, E “The Scattering of alpha and beta particles by Matter and the Structure of the Atom” Philosophical Magazine vol. 21 (1911) p. 671
[15] Bryson, Bill “A Short history of nearly everything” p. 141
/*54745756836*/